Part 4: Oxygen References
Context and Calculations
1. Introduction
Many of the subcategories in the Listed Activities legislation specify a reference oxygen concentration at which the emissions must be reported. For example, an oxygen reference of 11 % is specified in subcategory 5.8 below: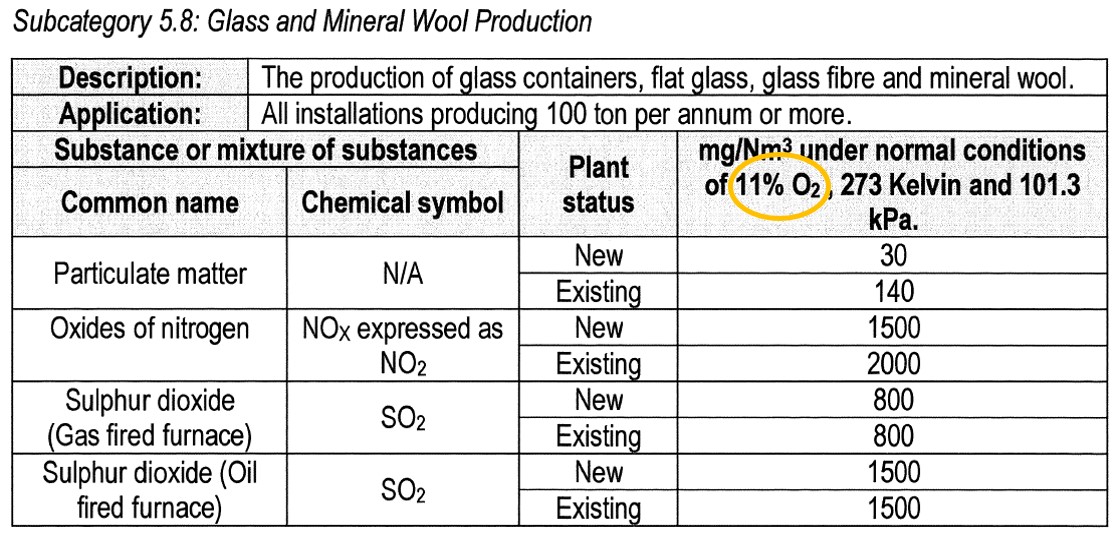
Reference oxygen concentrations are specified for combustion processes, such as the melting of glass in a furnace. Combustion is the process by which fuel burns in the presence of air. The concentration of oxygen in dry air is 21%, and this oxygen is consumed during combustion. The carbon and the hydrogen that are in the fuel combine with the oxygen to form carbon dioxide and water respectively, as per the formula beneath:
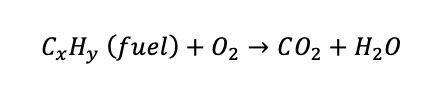
Because oxygen is consumed during combustion, the amount of oxygen that is in the stack gas, downstream of the furnace, contains less than 21% oxygen.
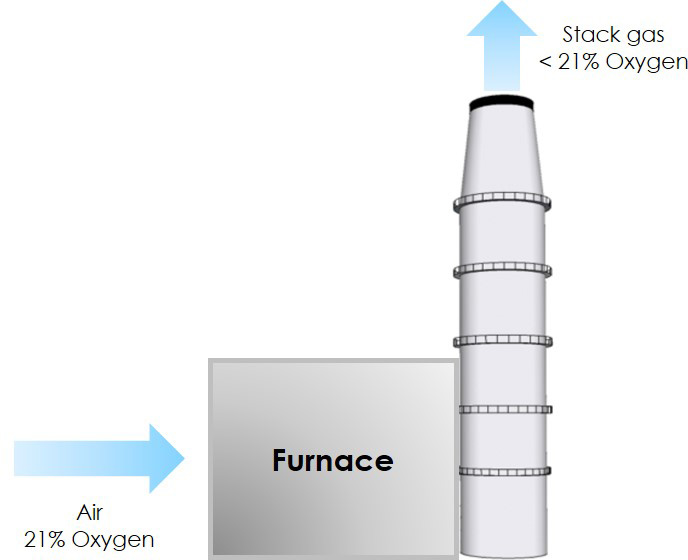
The fuel in the furnace requires a very specific amount of air to burn. This specific amount of air is referred to as the stoichiometric amount. If the stochiometric amount of air were provided to combust the fuel, then all of the oxygen would be consumed and there would be 0 % oxygen in the stack gas. However, combustion is imperfect, and it is always necessary to add more air than the stoichiometric amount to improve combustion. This additional air is referred to as excess air. It is important to provide just enough excess air for all of the fuel to be burned – but not too much excess air and not too little.
If too little excess air were supplied, not all of the fuel would be burned. The visible effect of this would be dark smoke from the chimney stack. If too much excess air were supplied, the additional air would cool the fire and carry heat out of the chimney stack – which is a waste of energy.
Because excess air is supplied, there will be oxygen left over that has not combusted. Therefore, more than 0 % oxygen will be measured in the stack gas. Different fuels combust differently and therefore require different amounts of excess air.
Solid fuels like coal and wood (biomass) require the most excess air. In a well operated coal fired furnace there may be 7% oxygen still remaining in the stack gas. In a well operated wood fired furnace there may be 10.5% oxygen still remaining in the stack gas. That is why the legislation often states 10% as the oxygen reference for solid fuels.
Solid fuels require the most excess air because they are comprised of large pieces and at any point in time only the outer surface of each piece is available for combustion. Furthermore, the oxygen struggles to reach the fuel surface in order to combust, because of a layer of air that is called the boundary layer, that surrounds each piece. These problems are overcome by providing more oxygen.
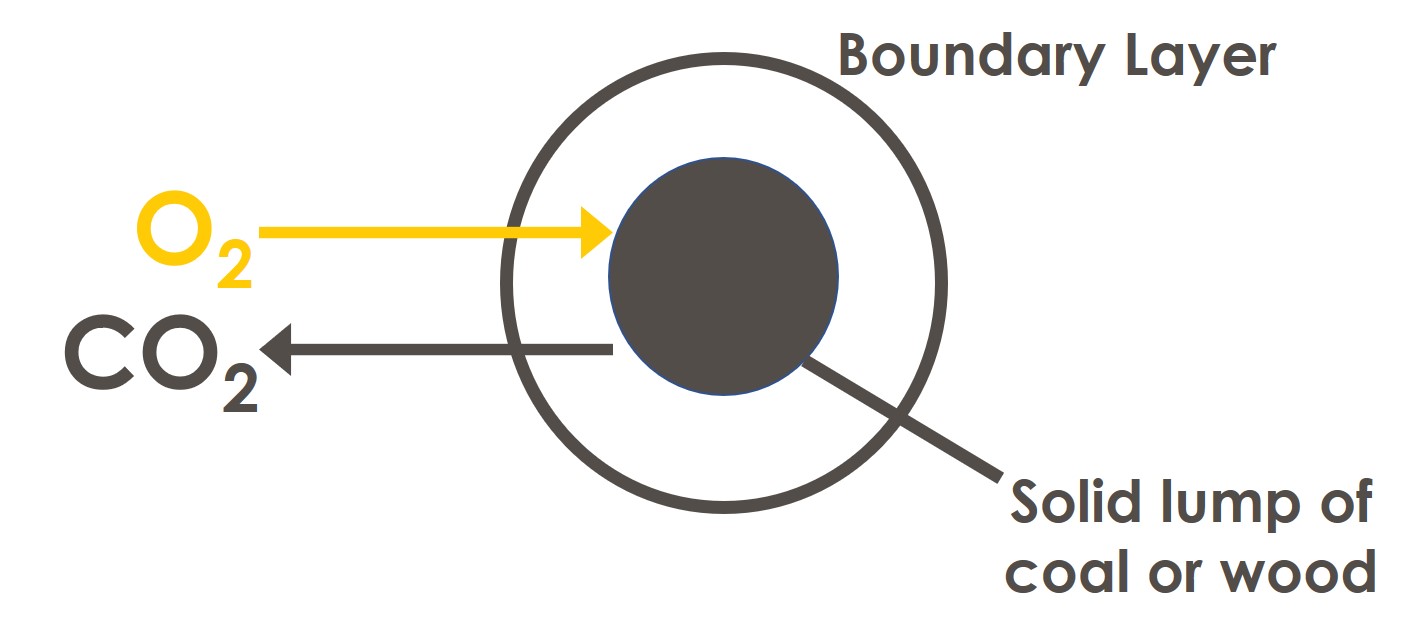
Liquid fuels require less excess air. This is because liquid fuels are sprayed (atomised) into the furnace as tiny droplets and the oxygen that is in the air can easily access each droplet. These droplets of liquid fuel have greater surface area than larger pieces of solid fuel:
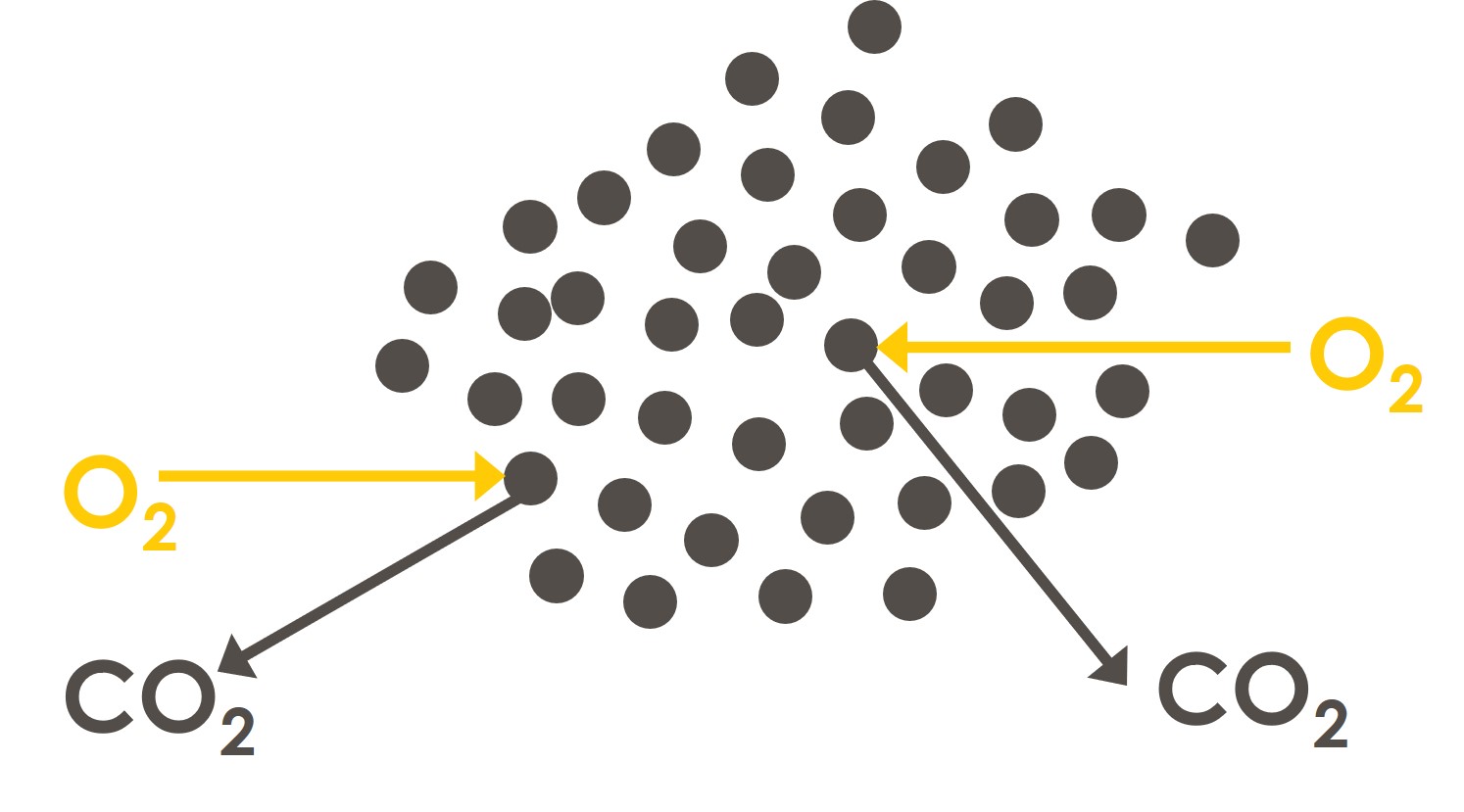
As such there is usually only 3% oxygen in the stack downstream of firing a liquid fuel.
Gaseous fuels mix almost perfectly with the air. Each molecule of gas can readily find a molecule of oxygen with which to react. Examples of gaseous fuels include methane, LNG, and LPG. Therefore, very little excess air is required when firing a gaseous fuel. One can measure as little as 1.8% oxygen downstream of the combustion of a gaseous fuel.
In most applications it is desirable to put as little excess air into the furnace as possible, because excess air carries heat out of the chimney stack. This heat was produced to be used in the manufacturing process (for example to melt glass). However, there are occasions where combustion takes place not to create process heat, but for other purposes. One of these purposes is the expansion inside of turbines. This is why much higher oxygen concentrations are measured downstream of turbines. The loss of heat through high volumes of excess air, is not of primary concern in this application.
Incinerators and cremators are another two examples where combustion does not take place with the exclusive purpose of creating process heat. Combustion takes place to destroy waste or organic matter. Again, the desired oxygen content in the chimney stack may differ in these applications.
2. Purpose of the Oxygen Reference
The emissions that exit the chimney stack can be diluted if the operator introduces more than the optimal amount of excess air into the furnace. This results in scientists measuring a lower concentration of pollutants. Diluted emissions misrepresent the actual emissions from the furnace.
To counteract dilution, the air quality legislation stipulates oxygen references for combustion processes. Reference oxygen concentrations are based on the typical oxygen concentrations that would be measured on the chimney stacks of these processes if these processes were operating normally. Boilers, furnaces, incinerators and turbines are examples of combustion processes that are regulated. Unfortunately, there is no solution to combat the dilution of emissions from non-combustion processes.
Table 1: Different Oxygen References for Combustion Processes (NEM:AQA)
The purpose of the oxygen reference is to reverse the effect of dilution so that the emissions that were measured on the stack are able to be compared with the limits in the legislation. There are 10 categories of Listed Activities, and 66 subcategories. 28 of these subcategories are combustion processes where an oxygen reference is applied (including the 2018 revision of subcategory 9.2).
3. Implementation
If an oxygen reference is specified in the legislation, then the emissions must be multiplied by the oxygen normalisation factor in order to reverse dilution and to calculate the final emissions:
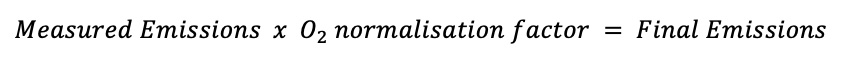
The oxygen normalisation factor is calculated as follows:

This factor allows the emissions to be reported just as they would have been had the dilution not occurred. Please watch the following video to see oxygen normalisations being performed:
4. Applicability
An oxygen reference can be applied whenever combustion occurs and depletes the oxygen in the stack gas. When no combustion takes place, no oxygen is consumed, and the oxygen concentration in the stack remains at 21 %. If one inserts 21 % into the formula the result is undefined because one cannot divide by zero:
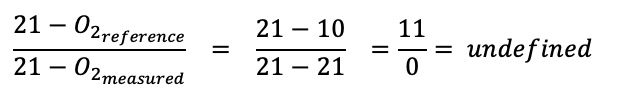
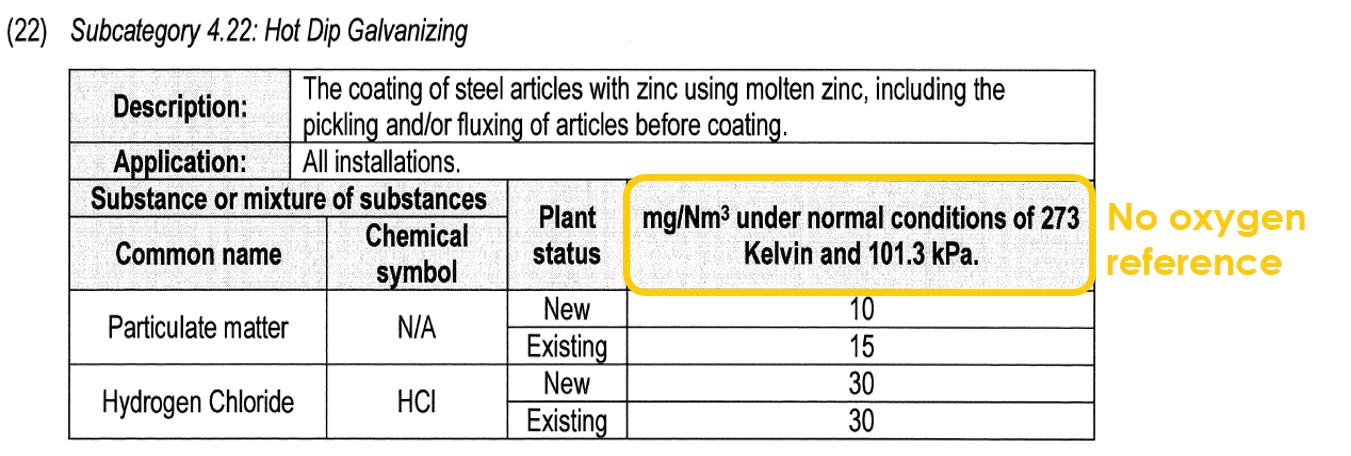
For this reason, processes that do not consume oxygen cannot be regulated with an oxygen reference. The example beneath shows this. It refers to Hot Dip Galvanising, where no combustion occurs:
Lastly, there are some processes that are indeed combustion processes, but which are not regulated with an oxygen reference. These are direct drying processes and include subcategories 4.1 and 5.2 of the listed activities:
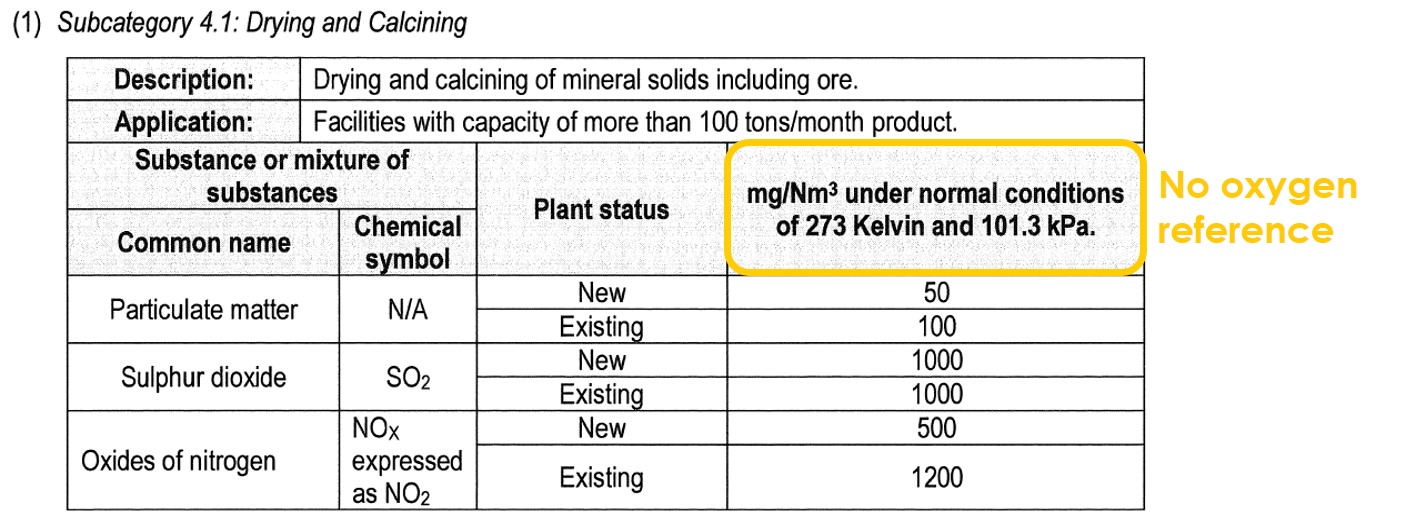
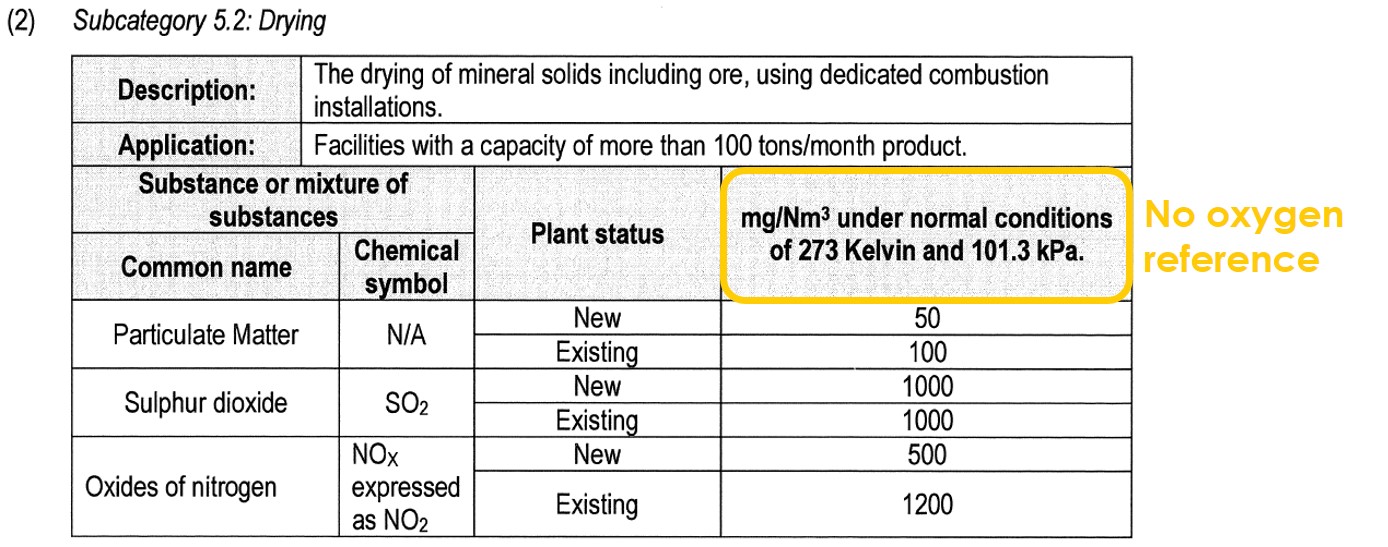
The reason that the legislation does not require an oxygen reference to be applied, is because the addition of dilution air after combustion is actually a process requirement. The dilution air is needed to lower the temperature of the hot combustion gases which could be as high as 800 – 1 000 ºC. When drying minerals such as sand, excessively high temperatures in the drier would melt (sinter) the sand. When drying wood, excessively high temperatures would burn the wood.
For these reasons, the hot combustion gases must be diluted with ambient air until they reach the optimal temperature to dry the wet material without burning or melting the material. For this reason, oxygen normalisations are seldom applied to drying processes.
5. Worked Example
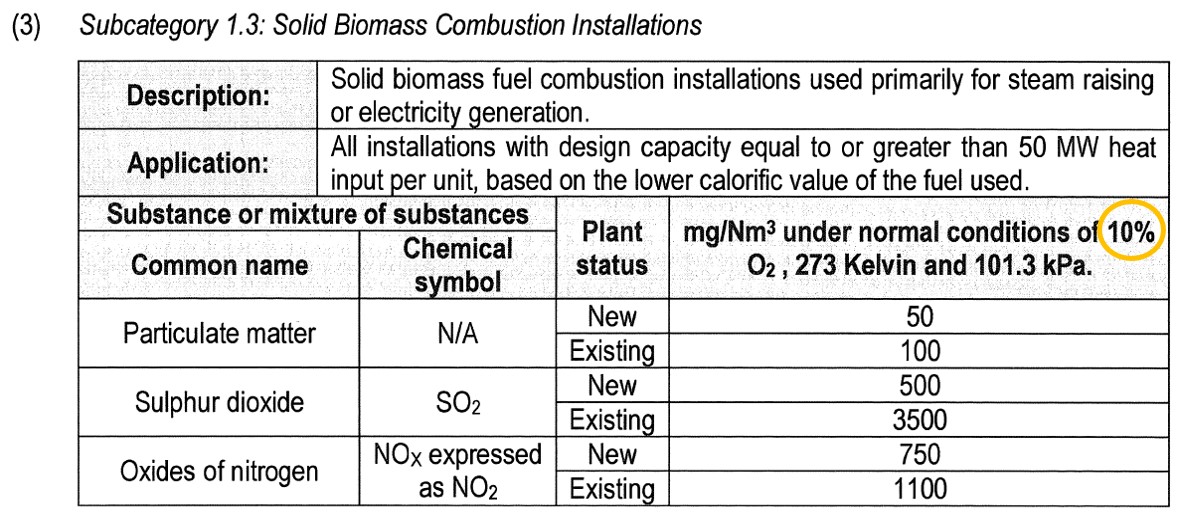
A sugar mill emits 300 mg/Nm3 of particulate matter at 16% oxygen from its boiler that is fired on bagasse. It is classified as subcategory 1.3 of the listed activities. The oxygen reference of this subcategory is 10% because a solid fuel is being burned. What is the normalised concentration of particulate matter after applying the oxygen reference?
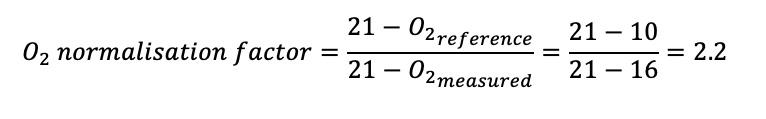
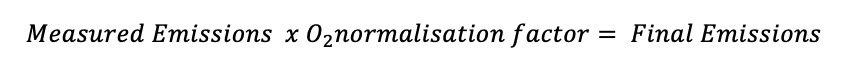
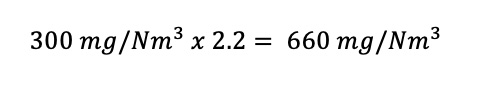
We are here to help, and our advice is always free of charge. Please feel free to contact us.
We love what we do.
